Dr Gareth Edwards is a Senior Bioassay Scientist at Cellomatics. In his blog, he discusses how, as an Oncology CRO, Cellomatics is developing spheroid models to provide new perspectives on research projects.
Cell culture is frequently employed in biological and biomedical research as it forms a convenient and easy model for studying processes involved in many different phenomena; for instance, tissue development, the method of action of toxins and carcinogens, how diseases such as cancer occur, and how potential new drugs are metabolised. This allows the steps in different biological signalling pathways to be elucidated and the method of action of potential drugs to be investigated.
Cells range from commercially available immortalised cell lines (including the famous HeLa cell line) to patient-derived ‘primary’ cells and, more recently, induced pluripotent stem cell models (these can be induced to differentiate into many different cell types). Furthermore, cells can be genetically engineered to express specific proteins of interest (potentially tagged with fluorescent or biochemical markers) or mutated proteins (to reflect diseases); genes can also be edited or knocked down using modern methods such as Crispr or siRNA. In recent years, the scale of experiments has moved from large plastic culture flasks to smaller multiwell plates, which enables the experimental scientist to simultaneously assay a range of conditions (including experimental repeats). Combined with multiplexed endpoints measuring cellular health or downstream responses to stimuli (where the output is based on fluorescence, luminescence and colourimetry and readings are automated), this has resulted in the development of high content/high throughput screening (HCS/HTS) which can generate large amounts of experimental data in a relatively short time.
Traditionally, in vitro cell culture models used in the laboratory have been based on 2-dimensional monolayers, where cells adhere to and grow on coated plastic surfaces in various items of culture-ware. But where in the body do cells grow on coated plastic surfaces, and how realistic a model for testing does this provide?
Whilst current monolayer models perform valiantly for studying the basic biology of mechanisms in isolation, how well do they replicate conditions within the body, where cells are bounded on all sides by other cells or biological matrices? In recent years, this idea has been taken on to develop a range of 3D culture models such as spheroids formed of a single cell type and organoids formed of multiple cell types which self-organise into organotypic mimetic structures (mimicking the tumour microenvironment)1,2,3. By receiving developmental and phenotypic cues from neighbouring cells rather than an artificial surface, these form a more realistic model for cell biologists to use to assess how genes and chemicals can modify the cellular phenotype4. As an example, focal adhesion kinase (FAK) plays a role in the generation of insulin-producing monohormonal pancreatic endocrine β cells from human pluripotent stem cells (hPSCs) as FAK phosphorylation was downregulated in 3D culture models thus promoting endocrine cell commitment, and treatment with a FAK inhibitor to mimic this in 2D cultures enhanced the expression of β cell-specific factors5. Therefore, the mode of growth of cells in culture and the interaction with their growth substrate may influence cellular phenotypes.
Multiple methods exist for generating 3D culture models. Cells can be cultured in ultra-low adhesion culture plates, where the lack of adhesion to the plate surface ‘encourages’ them to adhere to one another-the nascent cell aggregate rounds up and forms a rough ball of dividing and interacting cells6. Cells can be embedded in matrices, for instance agarose or Matrigel, whereby cells divide to form encapsulated balls of cells7. A more outlandish scheme to generate 3D aggregates of cells involved levitation in an ultrasound standing wave8, which demonstrated that the cytoskeleton of cells grown in 3D more closely mimics that seen in tissue samples than cells grown on a plastic surface9. These models show differing biochemistry to cells grown on traditional culture-ware, from differing drug responses10 to more rapidly acquiring specific phenotypic functions11. More recently, 3D printing techniques have been applied to generate organoids en masse with a higher degree of reproducibility using cells (both cell lines and patient-derived cells) and self-gelling matrix components which show upregulated genes involved in hypoxia, adhesion and EGFR signalling, downregulated cell cycle genes, and enhanced resistance to commonly used chemotherapeutic agents compared to 2D cultures12.
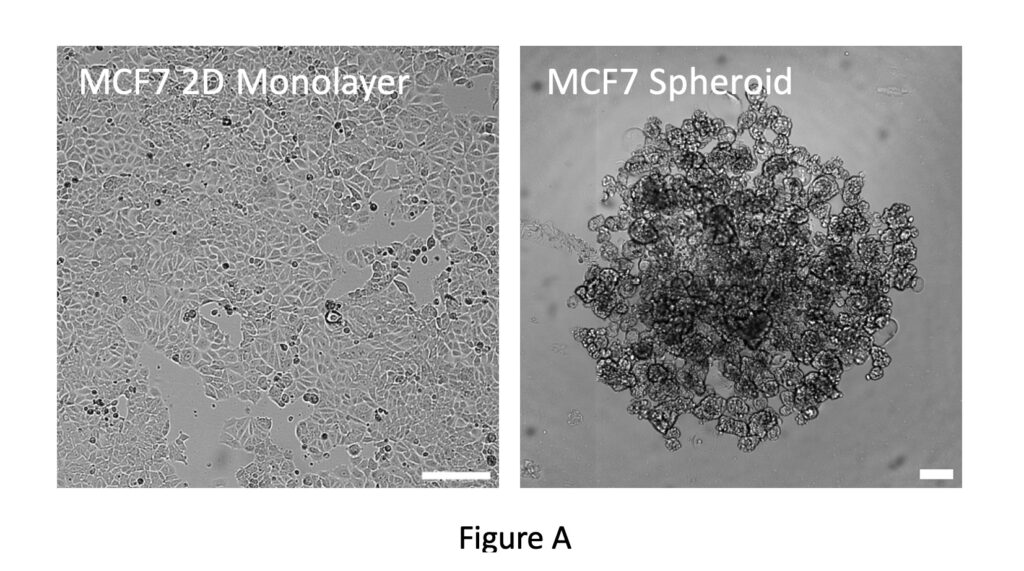
At Cellomatics (an Oncology CRO), we are keen to develop and exploit state of the art technologies in order to offer novel and exciting solutions to cater to the needs of our clients. To this end, we are currently developing spheroid models to provide new perspectives on research projects. Initial proof-of-principle experiments have been performed with the breast cancer cell line MCF7. These can be grown as a traditional 2D monolayer but when single-cell suspensions are plated out in an ultra-low-adhesion multiwell plate they coalesce to form a single spheroid in each well (Figure A).
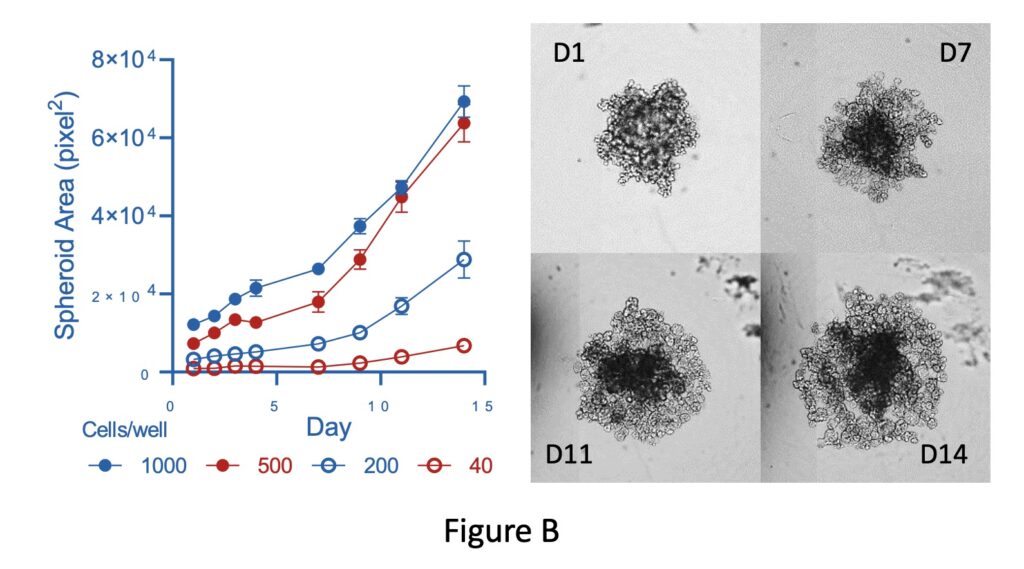
By using our JuliStage imaging platform, we can capture images for quantitative analysis-therefore, by plotting spheroid area against time we can analyse growth rates. This demonstrates that spheroid growth is determined by seeding density (Figure B).

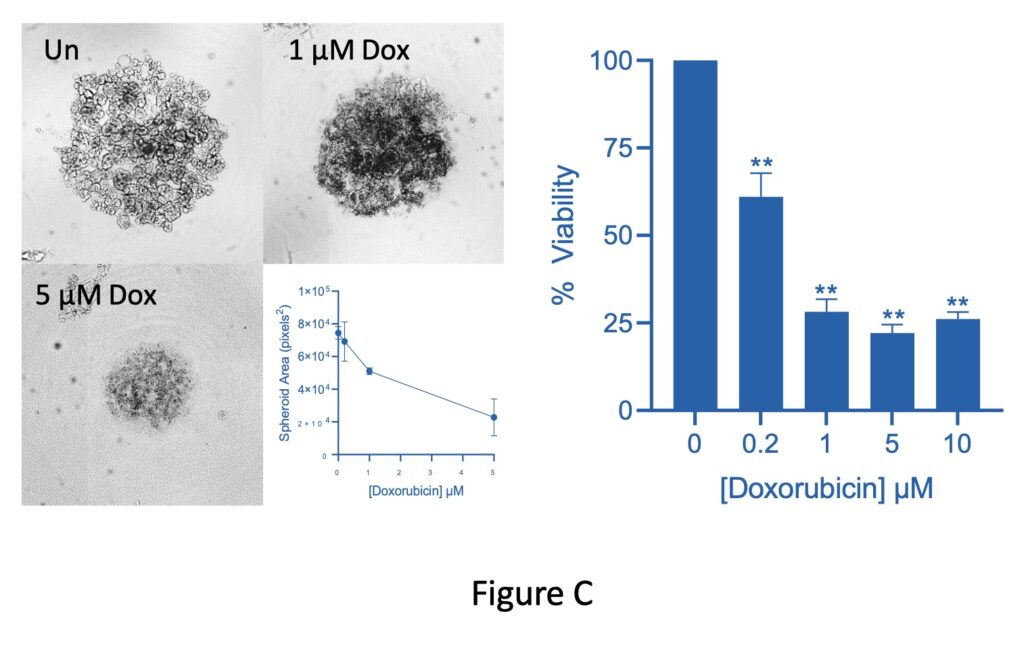
The 3D CellTiter Glo viability assay demonstrates that spheroids are comprised of viable cells. Exposure to toxic compounds such as Doxorubicin reduces spheroid area along with cell viability (Figure C).

The CellTiter Glo assay is a biochemical readout where ATP concentration is used to assess viability. As a proof of concept, this demonstrates that we can analyse the biochemical footprint of spheroids grown in a multiwell plate. Therefore, this platform can be used for high throughput screening of spheroid biochemistry in response to external stimuli. We are expanding the range of assays we can offer based on this platform, and progress on this will be discussed in a later blog entry.
Downstream from our work on spheroids, we are expanding our abilities in the field of immuno-oncology13,14. This is an exciting field of cancer research which involves harnessing the body’s own immune system to target cancerous cells. Over 250 clinical trials are currently in progress14 and novel immuno-oncological treatments are already proving to be successful15. Combination therapy involving nivolumab and ipilimubab which together function to allow immune cells to attack tumours was approved by NICE in June 202116. One area of interest is ADCC, or antibody-directed cell cytotoxicity13. Therapeutic antibodies bind to target cells, and this subsequently directs ‘effector’ cells from the immune system (expressing the FcγR receptor, and frequently NK cells) to attack and kill the cancer cells; reagents that can augment and enhance ADCC are of interest in addition to the antibodies themselves17. Trastuzumab targets breast cancer cells over-expressing Her2, subsequently binding to the receptor and slowing down cell proliferation, but may function additionally by inducing ADCC18. We are developing high throughput assays to assess ADCC which we will be able to offer to clients as a bespoke and highly customisable model. Routine cell culture coupled with high content screening is already in place and this provides a solid foundation for developing such an assay. However, this is not enough. We have the ambition to drive forwards the evolution of these models by enabling experiments to be performed in a 3D environment coupled with high-throughput screening. This will enhance the assessment of client-specified combinations of target and effector cells along with potential therapeutic antibodies and promoter reagents. As a result, the data generated will echo more closely the way that these cells interact in the body.
During work to set up these assays, a further advantage has become apparent. Screening using cell culture models can use a large amount of sterile, single-use plasticware. By moving to a 96-well multiwell plate format, we can potentially replace 96 individual tissue culture flasks with one small test plate, additionally reducing the volume of media and test reagents from 5ml to 0.2ml per replicate. Therefore, these techniques have the added benefit of reducing the amount of plastic and liquid waste we generate, thus increasing the environmental credentials of both Cellomatics Biosciences and our clients.
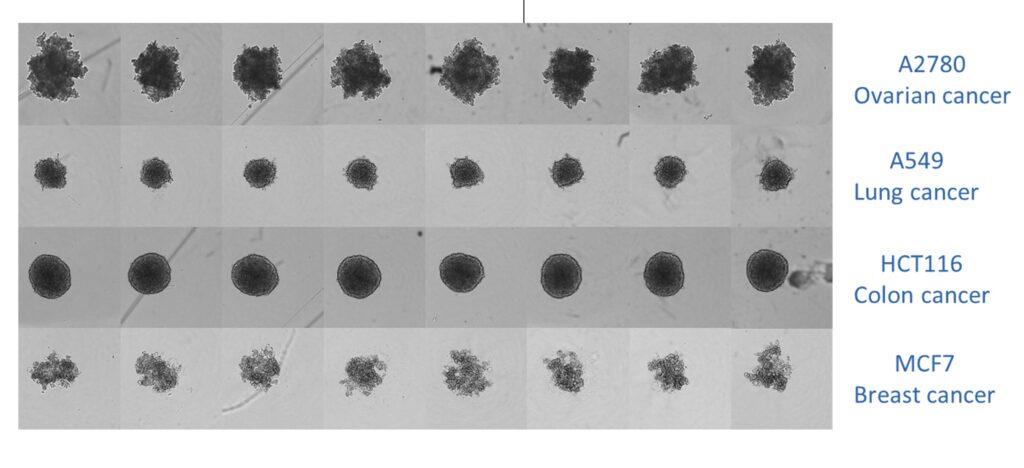

Combined image of spheroids grown in an ultra-low adherence 96-well U-bottomed plate. The image shows that shape and size are consistent for spheroids generated from each cell line.
Cellomatics is an oncology CRO, to find out how we can support your project, speak to a member of the team.
References
1Habanjar O, Diab-Assaf M, Caldefie-Chezet F, Delort L (2021). 3D Cell culture systems: Tumour application, advantages, and disadvantages. Int. J. Mol. Sci. 22:12200. DOI: 10.3390/ijms222212200. View link.
2Lee KH, Kim TH (2021). Recent advances in multicellular tumour spheroid generation for drug screening. Biosensors (Basel) 11: 445. DOI: 10.3390/bios11110445. View link.
3Cui X, Hartanto Y, Zhang H (2017). Advances in multicellular spheroids formation. J. R. Soc. Interface 14:20160877. DOI 10.1098/rsif.2016.0877. View link.
4Colombo E, Cattaneo MG (2021). Multicellular 3D models to study tumour-stroma interactions. Int. J. Mol. Sci. 22: 1633. DOI: 10.3390/ijms22041633. View link.
5Liu X, Qin J, Chang M, Wang S, Li Y, Pei X, Wang Y (2020). Enhanced differentiation of human pluripotent stem cells into pancreatic endocrine cells in 3D culture by inhibition of focal adhesion kinase. Stem Cell Res. Ther. 11:488 DOI: 10.1186/s13287-020-02003-z. View link.
6Madsen NH, Nielsen BS, Nhat SL, Skov S, Gad M, Larsen J (2021). Monocyte infiltration and differentiation in 3D multicellular spheroid cancer models. Pathogens 10:969. DOI: 10.3390/pathogens10080969. View link.
7Samimi H, Hohi AN, Irani S, Arefian E, Mhdiannasser M, Fallah P, Haghpanah V (2021). Alginate-based 3D cell culture technique to evaluate the half-maximal inhibitory concentration: an in vitro model of anticancer drug study for anaplastic thyroid carcinoma. Thyroid Res 14:27. DOI: 10.1186/s13044-021-00118-w. View link.
8Edwards GO, Bazou D, Kuznetsova LA, Coakley WT (2007). Cell adhesion dynamics and actin cytoskeleton reorganization in HepG3 cell aggregates. Cell. Commun. Adhes. 14:9-20. DOI: 10.1080/15419060701224849. View link.
9Edwards GO, Coakley WT, Ralphs JR, Archer, CW (2010). Modelling condensation and the initiation of chondrogenesis in chick wing bud mesenchymal cells levitated in an ultrasound trap. Eur. Cells. Mater. 19:1-12. DOI: 10.22203/ecm.v019a01. View link.
10Souza AG, Silva IBB, Campos-Fernandez E, Barcelos LS, Souza JB, Marangoni K, Goulart LR, Alonso-Goulart V (2018). Comparative assay of 2D and 3D cell culture models: proliferation, gene expression and anticancer drug response. Curr. Pharm. Des. 24:1689-94. DOI: 10.2174/1381612824666180404152304. View link.
11Liu J, Kuznetsova LA, Edwards GO, Xu J, Ma M, Purcell WM, Jackson SK, Coakley WT (2007). Functional three-dimensional HepG2 aggregate cultures generated from an ultrasound trap: comparison with HepG2 spheroids. J. Cell Biochem. 102:1180-9. DOI: 10.1002/jcb.21345. View link.
12Sbirkov Y, Molander D, Milet C, Bodurov I, Atanasov B, Penkov R, Belev N, Forraz N, McGuckin C, Sarafian V (2021). A colorectal 3D cancer 3D bioprinting workflow as a platform for disease modelling and chemotherapeutic screening. Front. Bioeng. Biotechnol. 9:755563. DOI: 10.3389/fbioe.2021.755563. View link.
13Fuertes MB, Domaica CI, Zwirner NW (2021). Leveraging NKG2D ligands in immuno-oncology. Front. Immunol. 12:713158. DOI: 10.3389/fimmu.2021.713158. View link.
14Liu H, Pan C, Song W, Liu D, Li Z, Zheng L (2021). Novel strategies for immuno-oncology breakthroughs with cell therapy. Biomark Res. 9:62. DOI 10.1186/s40364-021-00316-6. View link.
15Carter S, Thurston DE (2020). Immuno-oncology agents for cancer therapy. Pharmacol. J. 304:S2. DOI 10.1211/PJ.2020.20207825. View link.
17Rajasekaran N, Chester C, Yonezawa A, Zhao X, Kohrt HE (2015). Enhancement of antibody-dependent cell mediated cytotoxicity: a new era in cancer treatment. Immunotargets Ther. 4:91-100. DOI: 10.2147/ITT.S61292. View link.
18Boero S, Morabito A, Banelli B, Cardinali B, Dozin B, Lunardi G, Piccioli P, Lastraiolo S, Carosio R, Salvi S, Levaggi A, Poggio F, D’Alonzo A, Romani M, Del Mastro L, Poggi A, Pistillo MP (2015). Analysis of in vitro ADCC and clinical response to Trastuzumab: possible relevance of FcγRIIIA/FcγRIIA gene polymorphisms and Her-2 expression levels on breast cancer cell lines. J. Transl. Med. 13:324. DOI: 10.1186/s12967-015-0680-0. View link.